Abstract
Background: Hemophilia A (HA) is an inherited blood disorder caused by deficiency or dysfunction of coagulation factor VIII (FVIII). SPK-8011 is an adeno-associated viral (AAV) vector gene therapy targeting hepatocyte expression of FVIII, aiming to provide durable FVIII expression and long-term bleed prevention. A Phase I/II trial of SPK-8011 (NCT03003533/NCT03432520) has demonstrated multiyear sustained FVIII expression and notably reduced bleeding in people with HA, with no major safety concerns. There are challenges around preventing and managing cellular immune responses to transduced hepatocytes. Here, we present the outcomes of participants who received corticosteroids following vector administration to prevent and/or treat a presumed AAV capsid immune response.
Methods: In this open-label, multicenter, non-randomized, dose-escalation trial, people with HA were infused with SPK-8011 at one of four doses ranging from 5×1011 to 2×1012 vg/kg (full methods published in George et al. New Engl J Med 2021). Participants had baseline FVIII activity of ≤2% of normal levels and were negative for neutralizing antibodies to SPK200. Participants were monitored for 52 weeks after vector administration and enrolled in a 4-year long-term follow-up trial. Corticosteroids were used reactively at the investigators’ discretion upon a presumed capsid immune response, i.e., decrease in FVIII expression, increase in alanine aminotransferase (ALT) level, and/or positive peripheral blood mononuclear cells using interferon-γ enzyme-linked immunospot (ELISpot) to SPK200 peptides. Following loss of FVIII expression in two participants, a prophylactic regimen was explored. Assessments included duration of corticosteroid use and associated adverse events (AEs).
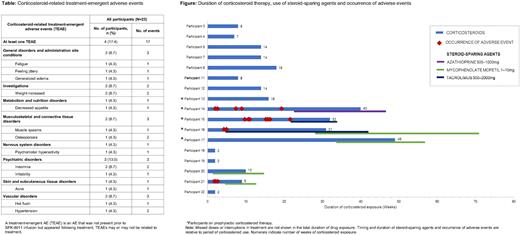
Results: Twenty-three males with HA were dosed with SPK-8011 and 17 received corticosteroids (12 reactively, 5 prophylactically); five did not have evidence of a capsid immune response, and one remains in the window of a capsid immune response in the follow-up period (data cut-off: 30 Jun 2022). The median (range) time to initiation of reactive corticosteroids was 9.3 (4.3-12.1) weeks post vector infusion in the 12 participants, with a median (range) duration of treatment of 8.5 (2-18) weeks. Two of these 12 participants completely lost FVIII expression following a presumed capsid immune response (one initiated emicizumab; one resumed FVIII prophylaxis); FVIII expression was maintained in the remaining 10 participants. Corticosteroids were administered prophylactically (2-4 weeks post vector) in five participants, with transgene expression maintained in all five. In four of these five participants, length of exposure was prolonged (31-49 weeks), with attempted corticosteroid tapering resulting in decreased FVIII expression, increased ALT and/or positive ELISpot assays outside the typical window of capsid immune responses. Four participants had AEs related to corticosteroid therapy (Table), with weight gain (8.7%; n=2) and insomnia (8.7%; n=2) being the most common. The occurrence of AEs attributed to corticosteroids increased with duration of use, particularly in those with exposure of >30 weeks. Prophylactic corticosteroids did not consistently prevent a presumed capsid immune response in these five participants and four experienced prolonged courses of corticosteroids necessitating steroid-sparing agents (Figure).
All five participants who received prophylactic corticosteroids and four participants who received reactive corticosteroids experienced a recurrence of one or more laboratory trigger(s) potentially linked to a presumed capsid immune response during their corticosteroid taper. Subsequent increased corticosteroid use did not consistently resolve these laboratory changes.
Conclusions: Overcoming the challenge of capsid immune responses to achieve long-term stable and predictable FVIII expression remains a goal of AAV gene transfer for HA. Reactive oral corticosteroids were well tolerated but did not prevent loss of FVIII expression in all participants. Early prophylactic corticosteroids did not consistently prevent presumed capsid immune responses and resulted in prolonged immunomodulatory courses with associated AEs. Data so far have permitted refinement of laboratory triggers and investigation is ongoing to identify an alternative, safe and optimal immunomodulatory regimen.
Disclosures
Evans:Penn State Health Milton S. Hershey Medical Center: Current Employment. Rybka:Spark Therapeutics: Consultancy; Merck: Research Funding; Spark Therapeutics: Membership on an entity's Board of Directors or advisory committees; Penn State Health: Current Employment. Croteau:Bayer, Pfizer, Sanofi, BioMarin, HemaBiologics: Consultancy; Spark Therapeutics, Genentech, Sanofi: Research Funding; Bayer, Pfizer: Honoraria; ATHN, Hemophilia Alliance, THSNA: Membership on an entity's Board of Directors or advisory committees. Tran:Sanofi: Research Funding; AstraZeneca, CSL Behrig: Honoraria; Pfizer, Takeda: Speakers Bureau. Rasko:Gilead, Roche, Novartis, Bluebird Bio, SPARK therapeutics, Cynata, Pfizer Inc: Consultancy; Genea: Current equity holder in publicly-traded company; Rarecyte: Current equity holder in private company. Jaworski:Spark Therapeutics: Current Employment. MacDougall:Roche: Current equity holder in publicly-traded company; Spark Therapeutics: Current Employment. Jaeger:BioNTech: Ended employment in the past 24 months; Spark Therapeutics: Current Employment. Mingozzi:American Society of Gene and Cell Therapy: Membership on an entity's Board of Directors or advisory committees; Roche pharma: Current equity holder in publicly-traded company; Spark Therapeutics: Current Employment. Chang:F. Hoffmann-La Roche Ltd: Current equity holder in publicly-traded company; Spark Therapeutics: Current Employment. Levy:Spark Therapeutics, Inc Board of Directors (Non-voting): Membership on an entity's Board of Directors or advisory committees; Roche Pharmaceuticals: Current equity holder in publicly-traded company; Genentech, Inc: Ended employment in the past 24 months; Spark Therapeutics Inc: Current Employment.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal